This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
Post-translational modifications
What are post-translational modifications?
When a protein is covalently modified it is considered a post-translational modification (because it happens after translation) [1]. There are over 200 characterized modifications. These help expand the function of mature eukaryote proteins, they alter the activity, localization, turnover, and interaction of proteins [1]. A few of the common post-translational modifications include: phosphorylation, glycosylation, lipidation, and protein cleavage.
What is protein phosphorylation?
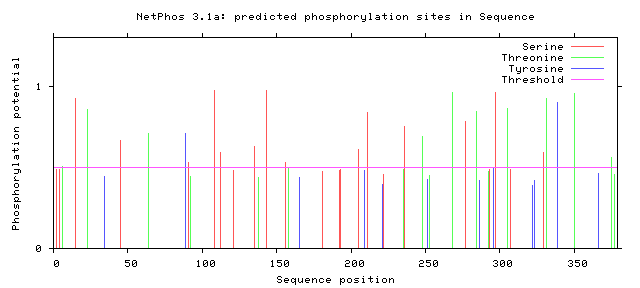
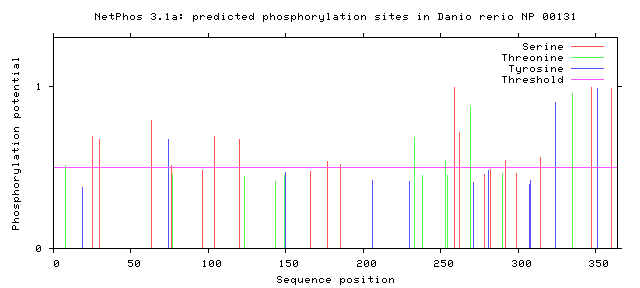
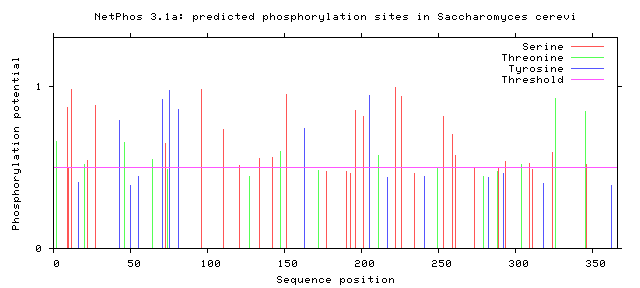
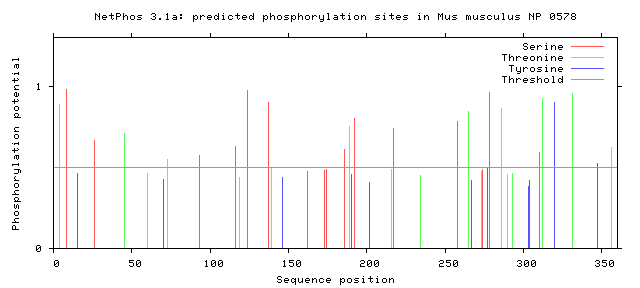
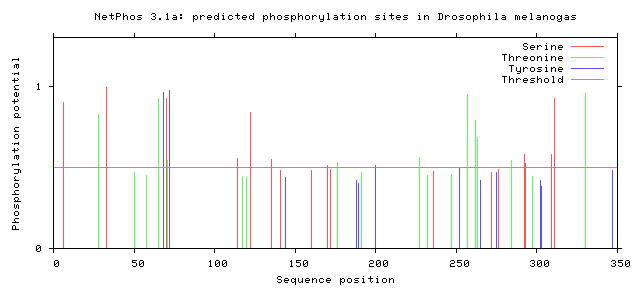
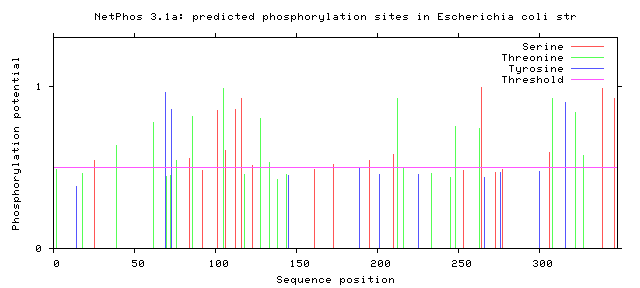
Phosphorylation is the addition of a phosphoryl group on proteins. These additions are temporary and help regulate the function of a protein, typically phosphorylation acts to active or deactivate a protein. The enzymes that phosphorylate proteins are called kinases and enzymes that dephosphorylate proteins are called phosphatases. The most commonly phosphorylated amino acid is serine, followed by threonine and then tyrosine. Below the the predicted phosphorylation sites of GALT are shown. Prediction of sites was done using NetPhos.
Are the predicted phosphorylation sites similar across species?
Conclusions
As shown above GALT has several phosphorylation sites along the entirety of its length. However whether theses sites are actually phosphorylated and biologically relevant is known. Further the sites appear to not be conserved across a broad range of species, which was to be expected. They are most similar between human and mice.